 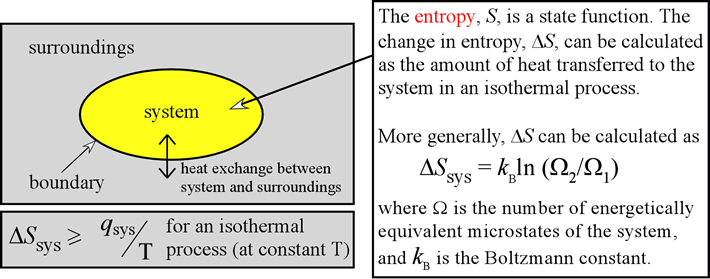
It will result in rigorous vibrations in solids and rapid translations in liquids and gases. Increasing the temperature will impact the motion of the particles. TemperatureĪccording to kinetic theory, a substance’s temperature is proportional to the particles’ average kinetic energy. The SI unit of entropy is J/K, and its dimension is. The symbol for entropy is S, and in its standard state, it is S˚. Thus, the change in entropy depends upon the initial and final state of the system, indicating that it is a state function. The change in entropy (ΔS) is the heat absorbed divided by the temperature. Where n is the number of moles and R is the universal gas constant. For a reversible, isothermal expansion from volume V 1 to volume V 2 at temperature T, the heat absorbed by the gas is given by Entropy can also be derived from thermodynamic quantities that are convenient to measure.Ĭonsider an ideal gas. However, calculating probabilities like W can be very challenging. Using statistical probability is very useful for visualizing how a process occurs. That is to say, doubling the number of molecules doubles the entropy. It is clear from this equation that entropy is an extensive property and depends on the number of molecules. The above equation is known as Boltzmann Equation, named after Austrian physicist Ludwig Boltzmann. W: Number of microstates corresponding to a given macrostate The key assumption made here is that each possible outcome is equally probable, leading to the following equation: Using Statistical Probability: Boltzmann Equation It can be quantitatively measured in terms of a system’s statistical probabilities or other thermodynamic quantities. How to Calculate EntropyĮntropy is a qualitative measure of how much the energy of atoms and molecules spreads during a process. These attributes of entropy are essential for formulating the Second Law of Thermodynamics. A positive entropy means an increase in disorder. Generally, the combined entropy of the system and the surrounding for a spontaneous process increases. Entropy and the Second Law of ThermodynamicsĪ system at equilibrium does not undergo an entropy change because no net change is occurring. Entropy is often called the arrow of time because matter tends to move from order to disorder in isolated systems. Since entropy measures disorder, a highly ordered system has low entropy, and a highly disordered one has high entropy. It is an extensive property, meaning entropy depends on the amount of matter. Entropy and the Second Law of ThermodynamicsĮntropy is a thermodynamic state function that measures the randomness or disorder of a system.The theory of probability is also discussed in the light of new theoretical findings related to the role of events, and also in terms of entropy and evolutionary thermodynamics. The evolutionary dynamics of complex systems, ranging from open physical and chemical systems (strange attractors, oscillating reactions, dissipative structures) to ecosystems have been investigated in terms of far from equilibrium thermodynamics (Prigogine). The relationship between entropy, as a non-state function, and the state function energy is stressed and discussed, in the light of the role of information. Entropy is discussed as a fundamental goal function in the far from equilibrium framework. Particular attention is devoted to goal functions, to relations of conceptualizations surrounding matter, energy, space and time, and to the interdisciplinary approach connecting thermodynamics and biology. This paper is an attempt to develop the new discipline of ecodynamics as a quest for evolutionary physics. WIT Transactions on State-of-the-art in Science and Engineering.WIT Transactions on Biomedicine and Health.WIT Transactions on Modelling and Simulation.WIT Transactions on Ecology and the Environment.WIT Transactions on Information and Communication Technologies.WIT Transactions on The Built Environment.WIT Transactions on Engineering Sciences.International Journal of Transport Development and Integration. 
International Journal of Sustainable Development and Planning.International Journal of Safety and Security Engineering.International Journal of Heritage Architecture.International Journal of Environmental Impacts.International Journal of Energy Production and Management.International Journal of Design & Nature and Ecodynamics.International Journal of Computational Methods and Experimental Measurements.Electrical Engineering & Electromagnetics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
